|
3/15/2024 0 Comments Entropy 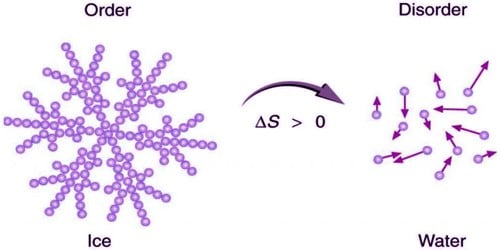
When a system's energy is defined as the sum of its "useful" energy, (e.g. 
Entropy is one of the factors that determines the free energy of the system. Quantitatively, entropy, symbolized by S, is defined by the differential quantity d S = δ Q / T, where δQ is the amount of heat absorbed in a reversible process in which the system goes from one state to another, and T is the absolute temperature. Entropy is an extensive state function that accounts for the effects of irreversibility in thermodynamic systems. In recent years, entropy has been interpreted in terms of the " dispersal" of energy. Entropy change has often been defined as a change to a more disordered state at a microscopic level. In contrast the first law of thermodynamics deals with the concept of energy, which is conserved. Spontaneous changes occur with an increase in entropy. The concept of entropy in thermodynamics is central to the second law of thermodynamics, which deals with physical processes and whether they occur spontaneously. Ice melting - classic example of entropy increasing described in 1862 by Rudolf Clausius as an increase in the disgregation of the molecules of the body of ice. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
